Daijiworld Media Network - Washington
Washington, Mar 26: A group of US senators pressed federal authorities to take swift action against the illegal online distribution of abortion medications, highlighting concerns about international supply chains, including those connected to India.
In a letter addressed to Food and Drug Administration Commissioner Martin Makary, the lawmakers urged immediate intervention to block the entry of what they described as “misbranded and unapproved” abortion drugs into the US. The medications mentioned—mifepristone and misoprostol—are typically used in regulated medical settings.
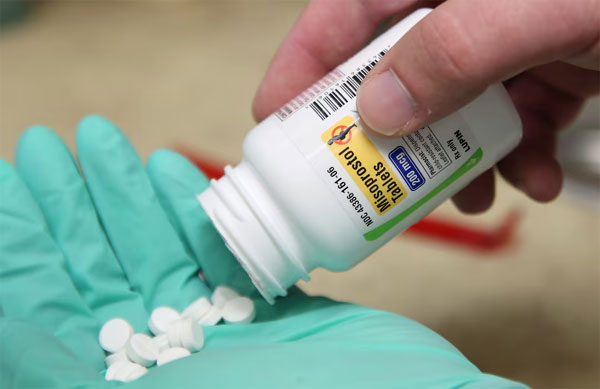
According to the senators, the unchecked online sale of these drugs not only puts women’s health at risk but also weakens the FDA’s mandate to safeguard consumers. They warned that the ease of purchasing such medications online, often without medical supervision, could lead to serious health complications and increase the likelihood of coercive or uninformed use.
The letter also shed light on the expanding global supply network behind these sales. It noted that many drugs purchased through foreign websites are sourced from manufacturers based in India. Lawmakers emphasized the rapid growth of this online market, citing cases where tens of thousands of packages were shipped annually across multiple US states, with estimates reaching up to 100,000 shipments in 2023 alone.
Four primary distribution channels were identified: US-based telehealth providers, overseas clinics, informal networks, and digital pharmacies. Many of these, the senators argued, operate outside proper regulatory frameworks and often dispense drugs without valid prescriptions.
They further alleged that some websites mislead consumers by claiming to offer FDA-approved products, while actually distributing unapproved versions produced abroad. Additional concerns were raised about certain platforms advising users to withhold information from healthcare providers after taking the drugs.
To address the issue, the lawmakers called on the FDA to intensify enforcement efforts. Their recommendations included issuing warning notices, targeting website domains involved in illegal sales, and strengthening coordination with customs and postal services to intercept shipments.
The letter also pushed for criminal investigations where necessary, referencing a past case in which a US resident faced prosecution for distributing abortion pills sourced from India without prescriptions.
Led by Bill Cassidy and supported by Steve Daines, James Lankford, Cindy Hyde-Smith, and Lindsey Graham, the group has asked the FDA to outline its enforcement strategy by April 8.
Medication-based abortion remains a common practice in the US when conducted under proper medical guidelines, typically involving a combination of mifepristone followed by misoprostol.